Poly(methyl vinyl ether-alt-maleic acid monoethyl ester) average Mw 130,000 LS, 50wt. ethanol 25087-06-3

Cationic Polymerization of Phenyl Vinyl Ethers: Investigations of the Propagation Mechanism, the Living Polymerization of Ortho-Substituted Derivatives, and the Step-Growth Polymerization of Divinyl Derivatives | Macromolecules

Living cationic polymerization of α‐methyl vinyl ethers using SnCl4 - Yonezumi - 2008 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

Explain why methyl vinyl ether (CH2=CHOCH3) is not a reactive dienophile in the Diels-Alder reaction. | Homework.Study.com
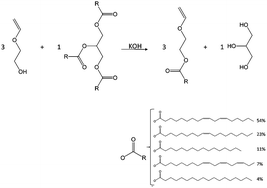
Living carbocationic polymerization of a vinyl ether monomer derived from soybean oil, 2-(vinyloxy)ethyl soyate - Green Chemistry (RSC Publishing)
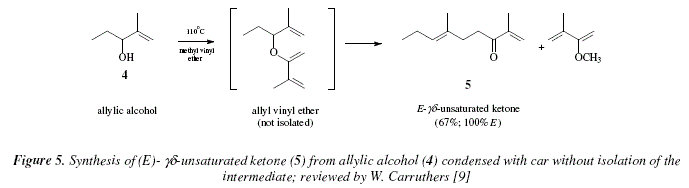
Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9

Recent advances in applications of vinyl ether monomers for precise synthesis of custom-tailored polymers - ScienceDirect

Organocatalyzed Controlled Copolymerization of Perfluorinated Vinyl Ethers and Unconjugated Monomers Driven by Light | ACS Catalysis



![Synthonix, Inc > benzyl vinyl ether - [B26636] Synthonix, Inc > benzyl vinyl ether - [B26636]](https://synthonix.com/images/product/26636.png)