
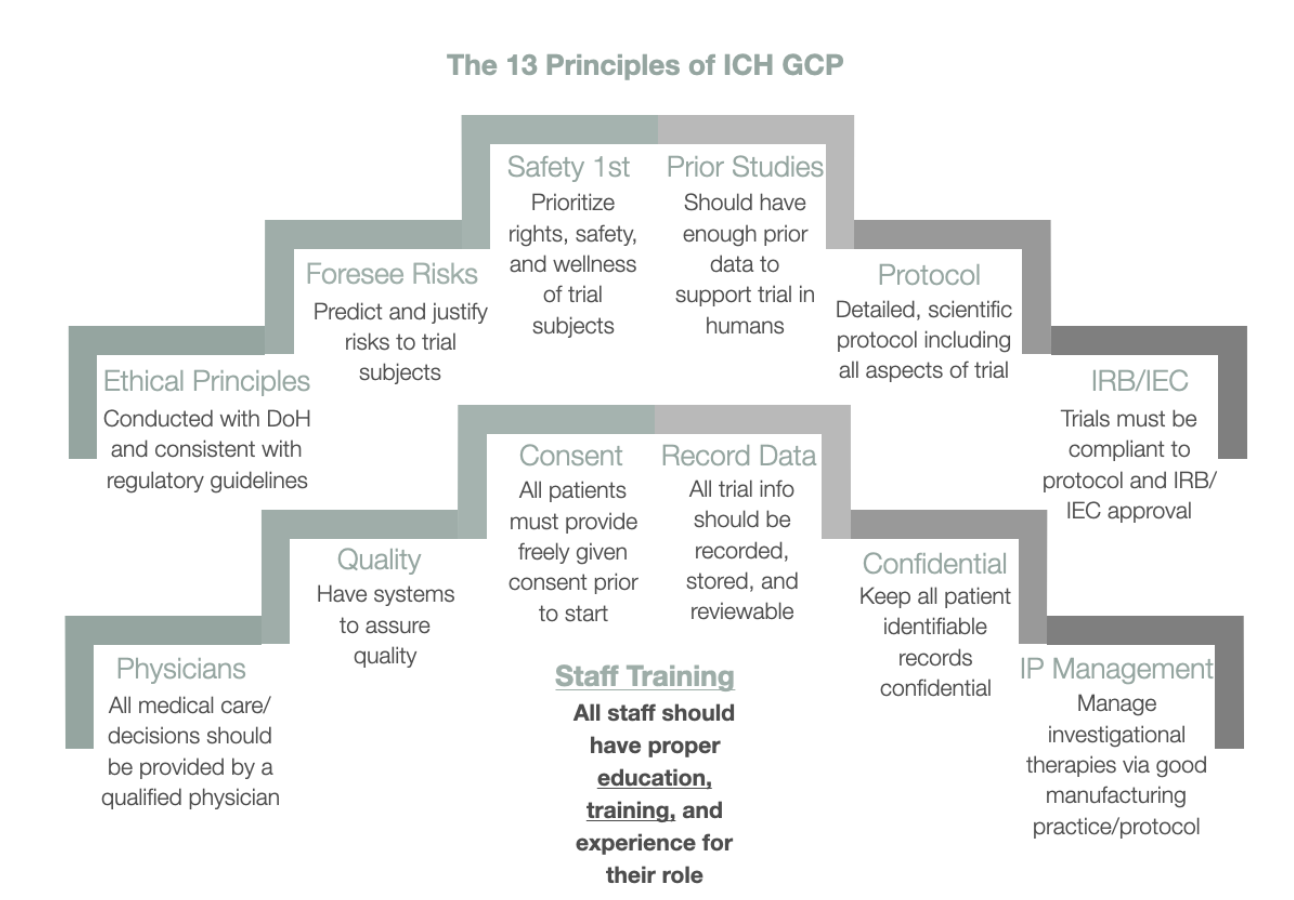
Book M1: 2022 Mini Pocket-Sized (3" x 5") ICH Guidelines for GCP (E6) – Clinical Research Resources, LLC

Consorzio per Valutazioni Biologiche e Farmacologiche - CVBF organises the “ ICH-Good Clinical Practice (GCP) Training Course”, an e-learning training course aimed at providing a guide for all individuals that are involved in
















(135).jpg)

