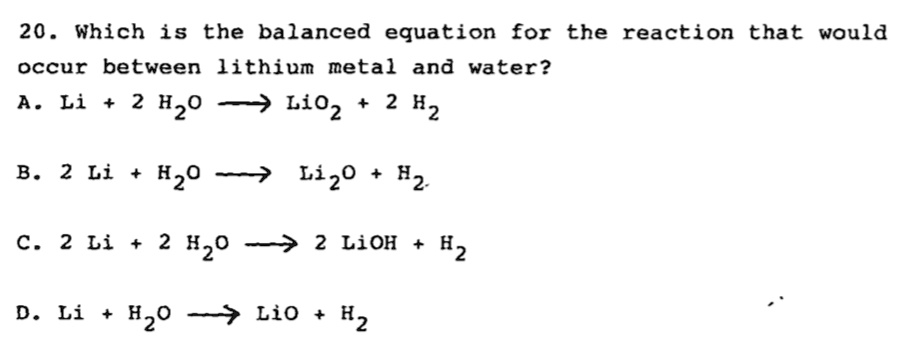![SOLVED: 20. Classify the following reaction: Li2O (s) + H2O (l) 2 LiOH (aq) [balanced] a)Combustion b)Single displacement c)Decomposition d)Combination (Synthesis) e)Double displacement SOLVED: 20. Classify the following reaction: Li2O (s) + H2O (l) 2 LiOH (aq) [balanced] a)Combustion b)Single displacement c)Decomposition d)Combination (Synthesis) e)Double displacement](https://cdn.numerade.com/ask_previews/29e63945-c838-4362-8ad3-2f5accabcc66_large.jpg)
SOLVED: 20. Classify the following reaction: Li2O (s) + H2O (l) 2 LiOH (aq) [balanced] a)Combustion b)Single displacement c)Decomposition d)Combination (Synthesis) e)Double displacement
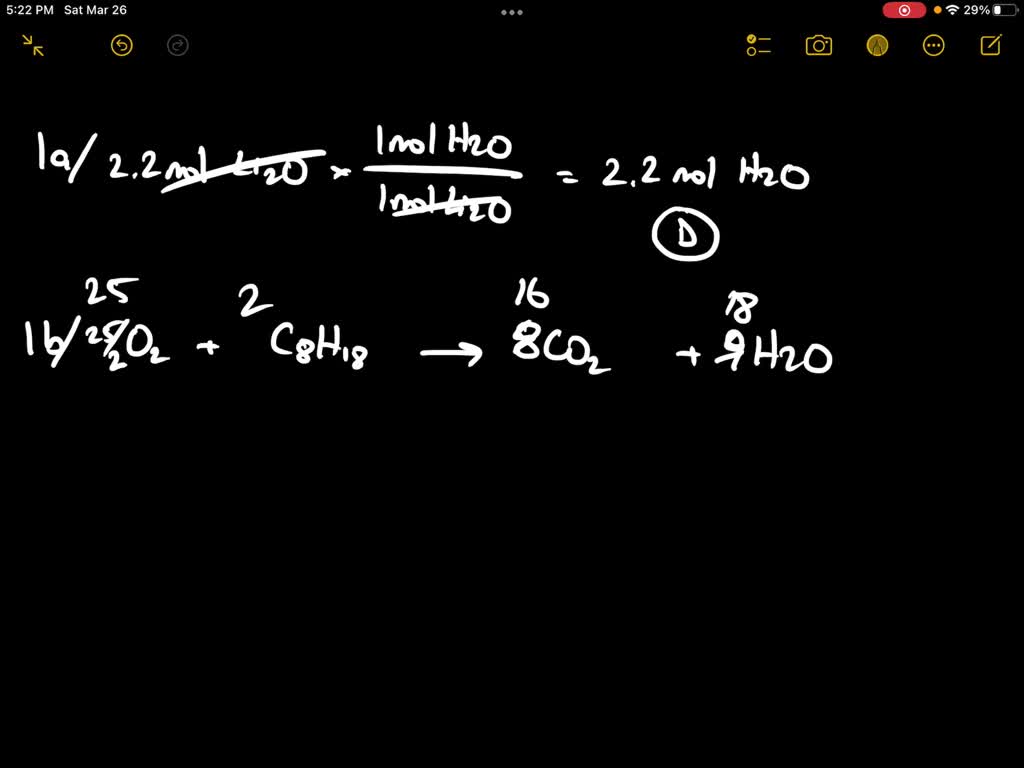
SOLVED: General Chemistry Please help me Q-1 a- How many moles of water are needed to react with 2.2 moles of Li2O? Given: Li2O + H2O → 2 LiOH Select one: A.

H2O Party on Instagram: “H2O'ers your FULL NYE LINEUP ⚡️💫🥳 Who's gonna turn your groove switch on?? Drop it in the comments 💃🙌 Get your tickets at:…”

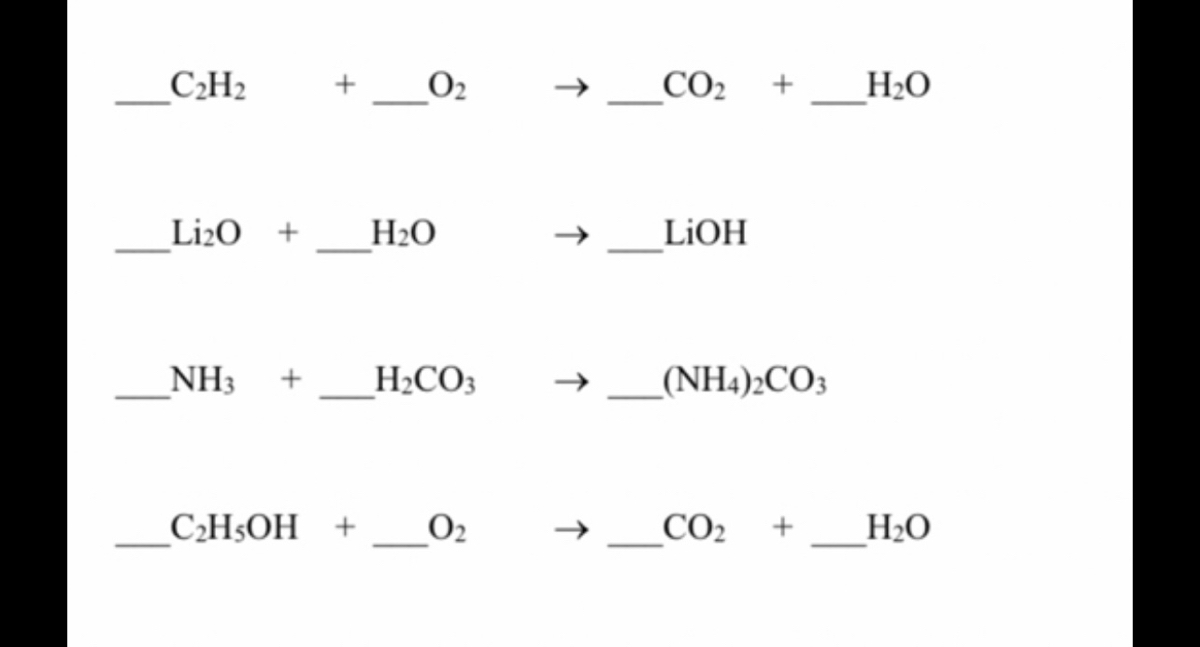
Reaction Types key - What Type of Reaction is it? Instructions: Balance the following equations and indicate the type of reaction. 1. Li2O H2O - | Course Hero

2LiOH overset("red hot")to Li2O + H2O, but 2NaOH overset("red hot")toxx Explain the above observation.