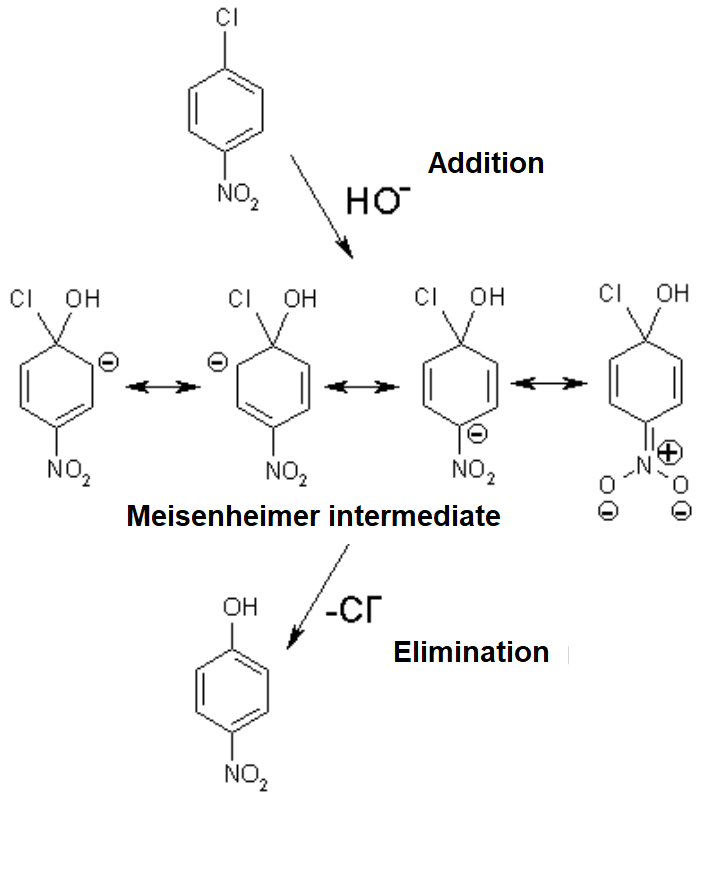
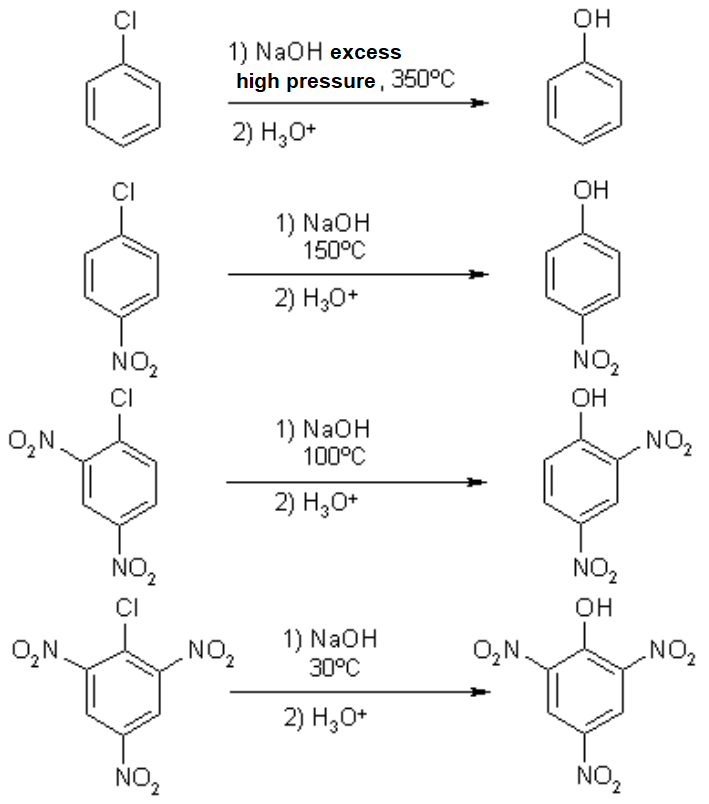
![PDF] The α-Effect in SNAr Reaction of 1-Fluoro-2,4-dinitrobenzene with Hydrazine: Ground-State Destabilization versus Transition-State Stabilization | Semantic Scholar PDF] The α-Effect in SNAr Reaction of 1-Fluoro-2,4-dinitrobenzene with Hydrazine: Ground-State Destabilization versus Transition-State Stabilization | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/cefe06be94f2a2f9cbcde1480f726f96da1f250b/2-Figure1-1.png)
PDF] The α-Effect in SNAr Reaction of 1-Fluoro-2,4-dinitrobenzene with Hydrazine: Ground-State Destabilization versus Transition-State Stabilization | Semantic Scholar

Cleavage of Aromatic C–O Bonds via Intramolecular SNAr Reaction and Preparation of P,C,Axial-Stereogenic Menthyl Phosphine Derivatives | Organic Letters
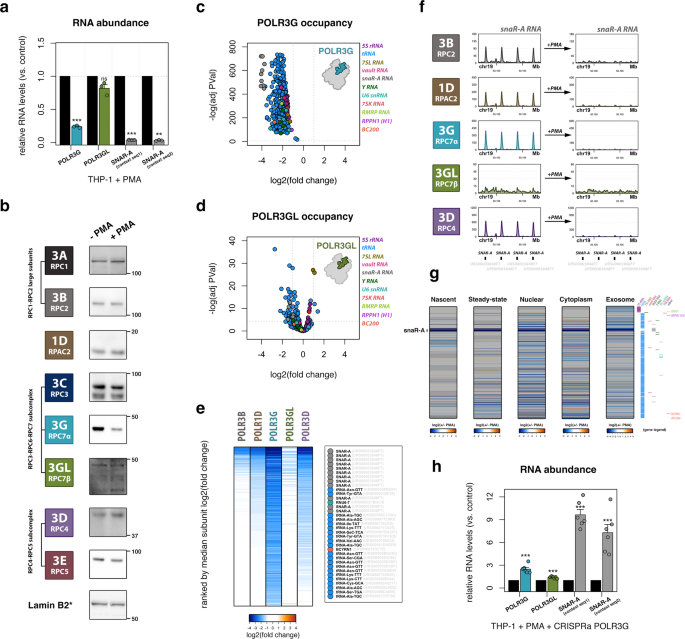
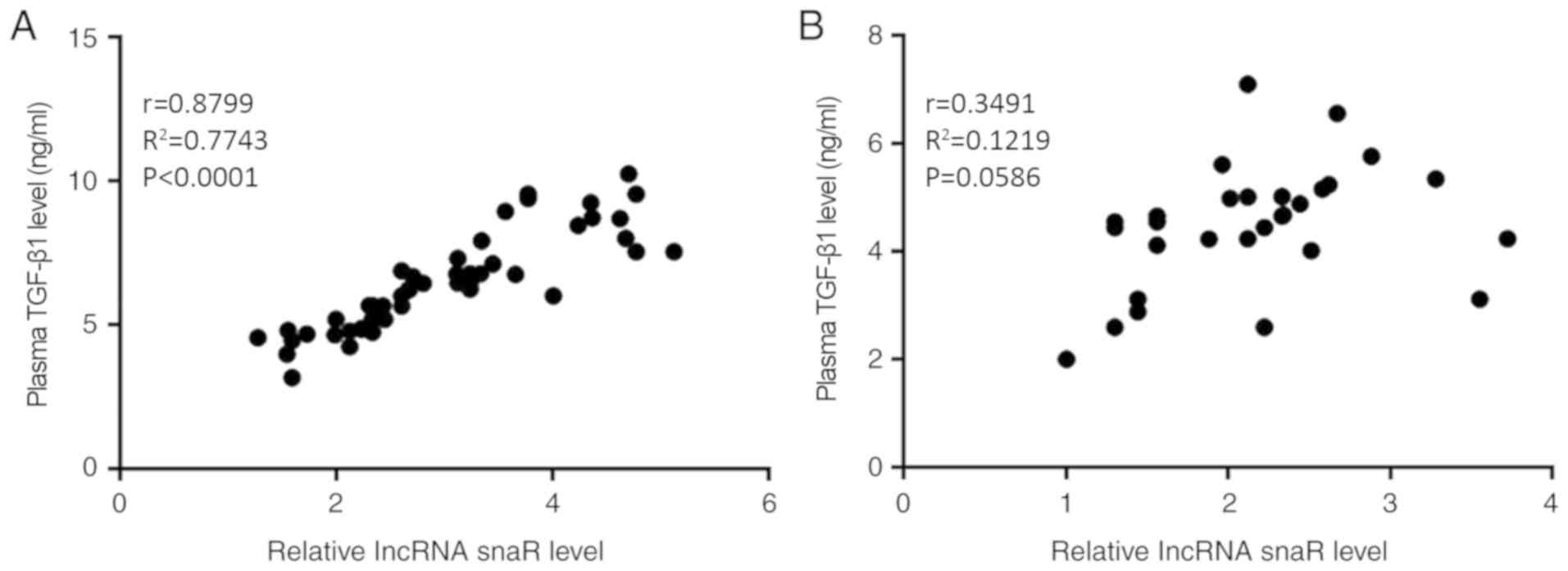
A cancer-associated RNA polymerase III identity drives robust transcription and expression of snaR-A noncoding RNA | Nature Communications

A cancer-associated RNA polymerase III identity drives robust transcription and expression of snaR-A noncoding RNA | Stanford Healthcare Innovation Lab

Aromatic Nucleophilic Substitution (SNAr) Reactions of 1,2- and 1,4-Halonitrobenzenes and 1,4-Dinitrobenzene with Carbanions in the Gas Phase - ScienceDirect

Douglas Carrel - The Gazoingial Snarp from whom Googly and Tilouga received the dreaded Pimpular Pea

Nucleophilic Aromatic Substitution (SNAr) as an Approach to Challenging Carbohydrate–Aryl Ethers | Organic Letters

Nucleophilic Aromatic Substitution (SNAr) and Related Reactions of Porphyrinoids: Mechanistic and Regiochemical Aspects - Sample - 2021 - European Journal of Organic Chemistry - Wiley Online Library

Aromatic Nucleophilic Substitution (SNAr) Reactions of 1,2- and 1,4-Halonitrobenzenes and 1,4-Dinitrobenzene with Carbanions in the Gas Phase - ScienceDirect

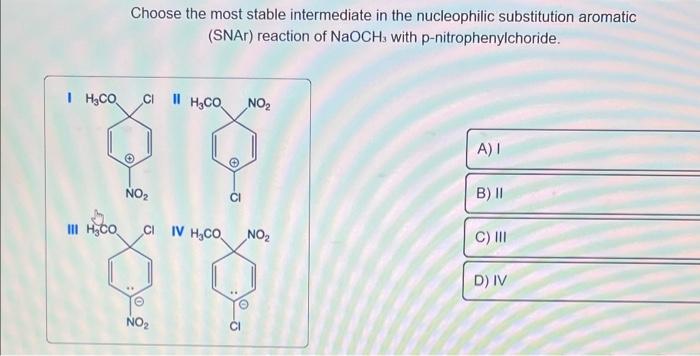
For the following reaction occuring through SNAr i.e. Substitution Nucleophilic aromatic reaction, the Meisenheimer complex is P , then the product Q will be:
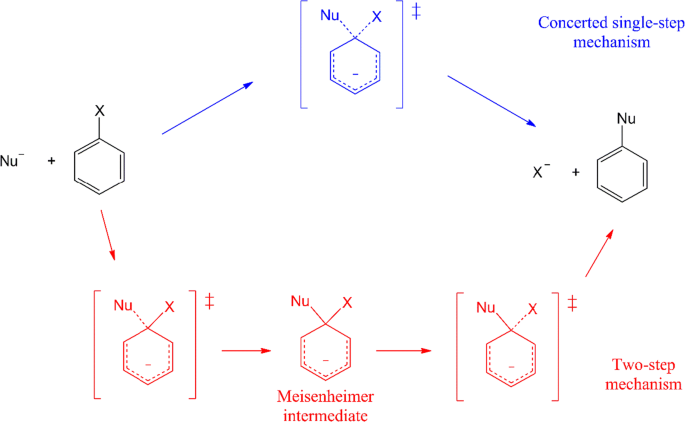
How difficult are anion-molecule SNAr reactions of unactivated arenes in the gas phase, dimethyl sulfoxide, and methanol solvents? | SpringerLink

Medium effect (water versus MeCN) on reactivity and reaction pathways for the SNAr reaction of 1-aryloxy-2,4-dinitrobenzenes with cyclic secondary amines

Aromatic Nucleophilic Substitution (SNAr) of meso-Nitroporphyrin with Azide and Amines as an Alternative Metal Catalyst Free Synthetic Approach To Obtain meso-N-Substituted Porphyrins | The Journal of Organic Chemistry

Nucleophilic Aromatic Substitution (SNAr) and Related Reactions of Porphyrinoids: Mechanistic and Regiochemical Aspects - Sample - 2021 - European Journal of Organic Chemistry - Wiley Online Library